What Is The Term For Surgical Repair Of Cartilage?
Initial degenerative changes in articular cartilage are typically silent. However, in one case symptoms appear, OA is a chronic progressive condition that causes pain and increasing disability [1,2]. Afterward accounting for mechanical disorders, OA has a high charge per unit of comorbidities (67%) in the general population, including diabetes and obesity [3]. Over the past decade, increasing interest in these comorbidities has led to Association of OA with cardiovascular disorders and hypertension [iii,4]. The term "metabolic syndrome" has been applied to the combination of obesity, diabetes, hypertension, and cardiovascular disease [4].
A study of the personnel in the Brazilian Navy institute xxx% of members had 2 or more risk factors (abdominal obesity, low HDL cholesterol, loftier fasting glycemia, high triglycerides, and loftier blood pressure) for metabolic syndrome [v]. In Finland, the loftier prevalence rate of metabolic syndrome in young obsess men decreased by 40% with the physical do required for war machine service [iv]. With such a loftier rate of comorbidities, OA is a serious public-health concern, and with a high numbers of practise injuries leading to posttraumatic OA, expenditures of OA related healthcare price frontwards to stable trips are rise. Repair of the damaged joint can have a profound touch on of improving mobility and overall fitness of an individual (Figure 1).
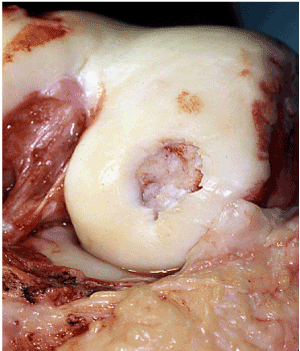
Figure 1. Damaged cartilage of femur
Molecular, mechanical, and traumatic events can all predispose a person for OA [6]. Genetic factors and the effects of aging can likewise play a role [1]. OA is considered disease of the whole joint, affecting all articulation tissues every bit they communicate with each other past releasing and responding to various signaling molecules [7]. Although the major molecules of articular cartilage are collagen and aggrecan, chondrocytes of the unique cells and cartilage that maintained joint hemostasis [2,vii]. Always typically characterized by degeneration of type 2 collagen, a deep pucker in the corporeality and size of aggrecan, and the macerated ability chondrocytes to remodel and repair the cartilage matrix [1]. With the disruption of blazon II collagen and loss of aggrecan, water content in the joint increases and the cartilage matrix loses tensile force [1]. When responding to inflammation in stress, osteoarthritic chondrocytes produce matrix degrading enzymes, which farther disrupt the joint matrix environment [7]. These molecular changes to the cartilage reduce the power of cartilage to disperse the load, minimize friction, and permit motility of the joint [8].
Other articulation factors too the cartilage also appeared to contribute to the osteoarthritic procedure. The elasticity of the synovial fluid, which lubricated to protect the knee articulation, decreases with historic period and is more than dramatic with OA [9]. The fluid of the knee joint is viscous, in part, because of the HA content; only in OA, the knee joint has nigh half the amount of HA as a healthy developed articulatio genus [nine]. Intuitively, was synovial fluid should crusade an increase in the friction coefficient [10], which would compromise the ability of the synovial fluid to lubricate and protect the articulation. Recently Caligaris and colleagues demonstrated that the friction coefficient of human tibiofemoral cartilage did non increase with increasing grades of OA, simply did increase with decreasing molecular weight of osteoarthritic synovial fluid [10]. From scientific viewpoint, they ended the intra-articular injections of loftier molecular weight synovial fluid may produce a statistically significant decrease in the cartilage friction coefficient [10].
Mechanical factors likewise contribute to OA through joint instability and misalignment [one]. Muscular weakness such equally from the quadriceps muscle can increase OA progression secondary to decreased articulation stability and the disability of the muscle to assistant shock absorption of the joint during gait. Military fibrillations require high activity levels at demand increased use of knees, placing troops at a loftier risk for any injuries to. The general population lifetime adventure of developing ever significantly higher for those with a history of knee injury (57%) compared with those without any injuries (42%) [11]. And a contempo report posterior medical way in the military, repairing colleagues noted that all warriors (100%) who sustaining injuries, later on developed OA [12].
Altered mechanics because of injury may be direct or indirect direct. Direct articular cartilage touch on can disrupt the knee meniscus, ligaments, cartilage, and bone [thirteen]. The long-term furnishings of cruciate and major meniscal injury are well known [13]. Even with surgical repair, a disrupted anterior cruciate ligament could not restore articulatio genus kinematics and articulation contact mechanics [14]. Because a military such equally physically agile population, the 10 times greater incident rate of anterior cruciate ligament injuries reported for military machine personnel compared with other non-military populations is not unexpected [xv]. Jones and colleagues reported that incidence for meniscal injury increased approximately 60% for every 5year age grouping to. In fact, approximately 50% of patients who underwent meniscal surgery developed articulatio genus OA within x-20 years [13].
Cellular changes back-trail with mechanical changes to the articulation that occur with injury [16]. Following sublethal injury to chondrocytes, the combination of apoptosis and mechanically driven shifts in chondrocyte phenotype leads to stress-induced signaling cascades inactivation of inflammatory mediators [7,16]. Other genes involved an injury response including protein kinases that promote apoptosis [16]. The joints response to injury will vary based on which jeans are activated, but the increased expression of inflammatory and apoptotic signals tin can lead to more rapid articular cartilage degeneration [7,16]. Thus, cellular responses to injury induced inflammatory responses that intern Perpetua cartilage degeneration and weaken the power to repair the joint [7]. These changes are probable to further contribute to the development of progression of OA [7].
To reduce the impact of post traumatic OA, current research efforts her examining means to preserve and protect the joint, to repair damaged articular cartilage, and to prevent jail cell death after injury [17].
Hurting from OA is a potent motivator for people to seek handling farther OA. Patients typically describe hurting that begins gradually and worsens over time. The pain in the articulation is not produced from the cartilage because cartilage does not have any nervus endings [18]. With motility, joint send signals to sensory fretfulness [ane]. In OA, the signaling is increased and is interpreted as hurting in the Fundamental nervous organization [one]. Discomfort from ever oftentimes hard to localize for patients. They may have OA and 1 portion of the joint, only perceive the pain radiating to the other side of the joint, or up and downwards the leg.
Synovial inflammation may further promote cartilage distraction or hinder the ability of cartilage to be repaired [7]. The resulting swelling tin exist very painful, and may decreased range of movement of the joint. Interestingly, this may be protective. Many patients will rest and reduce her activity level because of the swollen joint, which decreased potential impairment of the joint.
Developed articular cartilage on average experiences about 2 megapascals of force and the hip, knee, and talocrural joint [xix-22]. Meniscal or inductive cruciate injury can increase articular forces to well over 8 megapascals which is well across the normal load of 1-four megapascals which could explain a subsequent arthritic degeneration over months and years [23,24]. Articular cartilages viscoelastic, which in biology means a prior preoperative tissue that exhibits both mucilaginous and elastic behavior, (creep and stress relaxation); the cloth stress strain behavior depends on strain rate [25]. Articular cartilage is anisotropic, meaning the intrinsic textile properties depend on the matrix orientation and limerick as well as the direction in which the cartilages loaded. Split line patterns on the surface of a human femoral condyle using India ink stain reveals the orientation of the fibers and bear witness the anisotropic nature of articular cartilage that should be illustrated. Collagen restricts the swelling pressure of aggrecan and to only 20% of its capacity to associated water, which is 50 times its weight. The internal pressure within cartilage is 0.5 megapascals (75 pounds per square inch) [26].
The 4 zones of cartilage behaves somewhat differently with statically applied strains [27]. The highest axial strain measurements have been constitute in the transitional zone, nonetheless, this was significantly only with respect to the upper and lower radial zones. It is thought that this relatively low axial strain design in the lower radial zone is nigh likely related to high proteoglycan content of this zone and its attachment to the underlying subchondral bone [28]. A constant force we volition produce creep and the Deformation from the stressful increase with time and so long as the forces maintained.
Undersurface of articular cartilage, the collagen fibers have a tensile strength of 30 megapascals in the young developed. To understand there relevance of this figure, aluminum has a tensile strength of 70 megapascals and nylon has a tensile strength of 80 megapascals [29].
The concept of the relationship betwixt duration, intensity, frequency of an applied force on articular cartilage is described as an envelope of injury [30]. Single event traumatic episodes tend to produce lesions isolated to a geographic expanse and are nigh often associated with a specific injury event. In contrast, weather that develop slowly and are not associated with trauma are more likely to influence a wider area of the joint. These changes are associated with long-term biochemical changes and touch on a larger amount of cartilage effectually the infected site leading to osteoarthrosis [31]. Articular chondrocytes are by and large able to maintain cartilage throughout life by replacing lost or damage matrix that they freshly synthesized fabric. Synthetic activity is very well regulated and will rapidly increase to well above basal levels in response to cartilage injury. This suggests that synthesis activity is linked to matrix loss through some impairment control mechanism. A major stimulator of matrix synthesis is insulin similar growth fracture I (IGF-I). Its availability in cartilage is controlled by IGF bounden proteins (IGFBPs) that are secreted by chondrocytes. IGFBPs are office of a complex organization, terms the IGF-I access that tightly regulates IGF-I activity. The IGFBPs block IGF-I activity past sequestering IGF-I from its cell receptor. The binding poly peptide IGFBP-3 has recently been studied and found to increase with chondrocyte historic period paralleling an age-related decline in matrix synthesis activeness. IGFBP-3 is actually over expressed and osteoarthritic cartilage with resultant disturbances in metabolic action and subsequent matrix degeneration. These observations may indicate that IGFBP-three plays a crucial function and regulating matrix synthesis and cartilage. Cartilage damage command mechanisms may fail because of age-related changes in IGFBP-3 expression or distribution. It is probable that the chondrocyte and the expanse of territorial matrix play important roles and the cartilage impairment command past interfering with a chondrocytes ability to reply to IGF-i [32].
There are a multitude of biologic surgical techniques to address isolated traumatic articular cartilage defects. These techniques offering a 70 percent adept to first-class outcome in restoration of function and reduction in hurting (Figure 2).
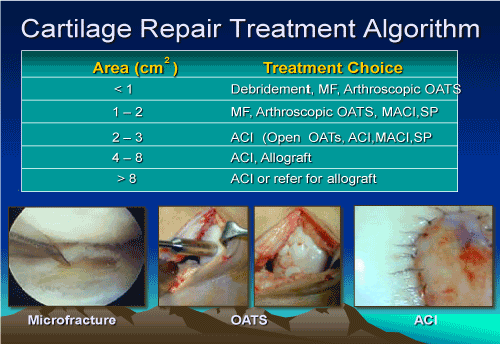
Figure 2. Treatment algorithm for cartilage repair
Early research has been directed at filling articular defects with tissue that approximates articular cartilage by stimulating the repair tissue from the marrow. Stem cells are capable of developing into a multitude of tissues. Bone, musculus, fat, tendon, and cartilage have all been obtained from these multipotent cells. A stem cell population capable of differentiating into cells that can produce cartilage are obtainable from the bone marrow. In human studies, the fraction of stalk cells that retained the ability to develop various types of tissue declines radically from ane in 20,000 cells and juvenile human being still but 1 in 4,000,000 during tardily adult life [33-37].
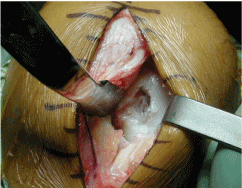
Microfracture of the subchondral bone is felt to evangelize stem cells from the bone to the articular surface resulting in nechondrogenesis [38,39]. The majority of studies are based on human clinical results from microfracture (Effigy 3).
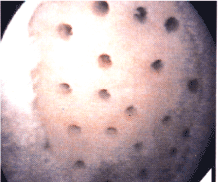
Figure 3. Microfracture of exposed os
The origin and differentiation of cells in the repair of 3 mm diameter cylindrical full-thickness articular cartilage and bone defects was studied past Shapiroet al. in New Zealand White rabbits [40]. In the kickoff few days, fibrinous arcades were established and the defects within the clot from surface edge to surface edge. At 10 days, Safranin-O staining revealed evidence of GAG-containing extracellular matrix. By 14 days, a type of cartilage like tissue was present immediately below the surface of the fibrinous arcades. This substance was densely populated with flattened fibrocartilaginous cells. Past 3 weeks, the sites of almost all the defects had a well demarcated layer of cartilage-like tissue. Autoradiography afterward labeling with 3H - thymidine and 3H - cytidine showed that chondrocytes from the adjacent native cartilage did non participate in the repopulation of the defect. The cartilage and but repair seemed to be mediated by the proliferation and differentiation of mesenchymal cells from the bone marrow. Interestingly, the radioisotope label which was taken up past undifferentiated mesenchymal cells appeared not only in the new chondrocytes but also in fibroblasts and osteoblast. Early traces of degeneration of the new cartilage matrix were seen in many of the defects by 12 weeks with the prevalence and severity of the degeneration increasing at 24 to 48 weeks. Polarized low-cal microscopy showed that the newly synthesize matrix had failed to integrate with the native cartilage adjacent to the drilled hole. Information technology was hypothesized that the lack of integration immune movement of the repair substance with the resultant early degeneration.
It has been shown that periosteal and os marrow drive cells tin produce similar design of differentiation intra-articular cartilage in New Zealand White Rabbits [41]. Trypsin was used to digestive costless periosteal and bone marrow bulldoze cells which were then implanted into a type I cartilage gel implanted into the site. Gel simply and control rabbits did not accept the same repair of cartilage and subchondral os full within the 3 mm defects as the cell impregnated gel defects. The cell impregnated gels had good reparative tissue that resembles Hylan cartilage. This Hylan like cartilage thinned out over a menstruum of 24 weeks [41] (Effigy 4).

Figure 4. Histologic view of microfracture
The successful transplantation of articular cartilage that is capable of restoring joint part, providing pain relief, and giving long-term reproducible first-class outcomes has been the holy grail of cartilage repair. Lexer attempted commutation of whole joints as far back as 1908 past 1925 was noted that even though whole joint transportation her initial good clinical results, the one that did non neglect went on to frank degeneration changes in the cartilage [42,43]. It was not until the early 1950s with a canine brute model was used to study morphologic and histologic differences between autografts and allograft afterwards transplantation. They must remain viable and capable of continued production of normal matrix. The matrix must remain mechanically competent without degeneration or exchange to a fibrocartilage. The bony portion of the graft must integrate with a course a mechanically audio matrimony with the host recipient site. Mechanical instability seems to signal the development of an immune response [44].
In the late 1970s and early on 1980s, there were a number of studies that examined various aspects of cartilage transplantation and animals to delineate the optimal conditions of transplantation. Autograft and allograft osteochondral transplantation were studied using gross observation, radiology, histology, histochemistry, and biochemistry. Lane and Brighton [45] examined articulation resurfacing in the New Zealand White rabbit using autologous transplanted femoral condyles. The lateral femoral condyle was completely excised and then fixed back into its original position with a straight Keith needle. Subsequent exam revealed that an lxxx% of these cases the cartilage surface appeared normal. At 1 year, histologic examination show that the thickness of the cartilage had been preserved with the exception of a few areas of localized thinning of the cartilage. Biochemical analysis of the harvested implants revealed a Dna, hydroxproline content, and S04 incorporation were equal to that of the non-harvested portions of the joint or to sham operated intact condyles.
Lipton et al. used a similar protocol to study follow transplantation of osteochondral grafts [46]. Osteochondral femoral allografts were performed and male adult New Zealand weight rabbits. Paired arthrotomies were performed simultaneously in dissimilar rabbits. The left lateral femoral condyles were osteomized and an osteochondral graft was harvested with 5 mm of subchondral bone. Care was taken to not transplant any soft tissue. The grafts were and so switched and translated from 1 rapid to the other and fixed with small K wires. The rabbits were allowed full weight bearing without immobilization. The condyles were and then harvested at three, half dozen, in 18 months. Physical test of the joints before harvest revealed no swelling, inflammation, or instability of any of the knee joints. However, gross inspection of the joints at necropsy revealed that one third of the joints had sustained advanced cartilaginous degeneration with fraying and even absent articular cartilage leading to eburnation of the bone. Two thirds of the joints appeared normal. Arthritic degeneration was noted to correlate with the joints that had not maintained an anatomic reduction later allograft transfer. Histologic analysis of the allografts that had maintained anatomic reduction revealed no differences when compared with cartilage from the undisturbed contralateral joint. Biochemical testing performed on the anatomically reduced allografts at 3, 6, in 18 months subsequently transplantation showed no statistical difference with respect to protein, collagen, hexosamin, or hydroxproline content. The incorporation of SO4 was studied both in vivo and in vitro and no statistically meaning differences were noted between the experimental and control condyles indicating acute metabolism was occurring. Biomechanical testing of the harvested specimens that had retained anatomic reduction revealed that there was no statistically significant difference in percent pitter-patter, instantaneous shear modulus, or relaxed shear modulus between allograft and control cartilage. The allografts that did neglect seemed to exercise so because of mechanical problems of fixation and no graft rejection [46].
In 1980, De Nubile et al. published data on aloe transplantation of preserved and fresh articular cartilage using an osteochondral plug model [47]. Information technology was thought the plug geometry would be mechanically stable construct and that the grafts would not undergo premature degeneration. Osteochondral plugs measuring 3-5 mm in bore were obtained with a remnant thickness of 1 mm of subchondral os from the New Zealand weight rabbits. A recipient rabbit was prepared and the donor plug was printing-fit into place. No boosted fixation too the press-fit was used for graft stabilization. The allografts transplanted in this matter were examined at iii, 6, in eighteen months. Gross exam revealed that there was nigh complete preservation of cartilage thickness in the majority of the specimens the edge his were well united with no clefts. Safranin - O staining revealed that the protocol similar a content of the allograft was preserved. A few of the plugs failed to unite to the recipient host bone and there was a proliferation of fibrous cloth around the plug. Some of the plugs also appeared to have subsided or were left protruding. The cases in which at that place was evidence of mechanical instability or subsidence or protrusion resulted in poor preservation of the cartilage matrix. Of special note, a few of the specimen samples appeared to accept developed an immediate neurologic reaction that was interpreted equally rejection. There were areas effectually these plugs that contained large numbers of monocytic and plasma inflammatory cells. The office felt that overall skillful to excellent results had been accomplished in 60% of the specimens. Those plugs or mechanically unstable seem to manifest this inflammatory response.
In 1991, Lazlo Hangodyet al., conceived of a method to use small-scale cylindrical bottle obvious osteochondral plugs for the treatment of focal chondral and osteochondral defects of the femoral condyles and the knee [48,49]. He tested the concept of mosaicplasty osteochondral transplant and debridement of working dogs. The graft sizes ranged from 2.7 to 4.5 mm. The peripheral margin of the distal femoral supracondylar ridge was selected equally the donor site because of its the cartilage morphology and non-weight begetting location. At iv weeks, the cancellous bone between the donor plugs and the recipient graft site had united. At 8 weeks, there was a seal betwixt the recipient and the donor surface that was interpreted to exist a fibrocartilage the produce matrix integration. The original donor sites filled with cancellous bone and fibrocartilage. This fibrocartilage was also firmly adherent to the surrounding cartilage. Histologic examination of the grafts revealed that the Highland morphology of both the chondrocytes in the cartilage matrix was maintained. In another written report, the grafts were all four.5 mm and eighteen were on the non-weight bearing lateral side and eighteen were on the weight bearing medial side. The lateral side fared better over a years' time and there was sustained proficient interplug fibrocartilage fill (hagody). On the basis of these results, the technique was then taken to the clinic and used on humans (Figures v and six).
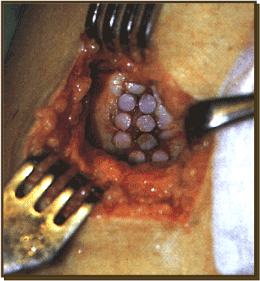
Figure five. Mosaicplasty
Effigy 6. Repair tissue from microfracture
Information technology is useful to compare organ allografts from other parts of the trunk to understand the considerations for osteochondral allograft surgery. In the instance of the middle transplant, the cells must remain alive for part and in that location is no tissue replacement from the recipient. The matrix is not replaced and it is maintained past the original organ cells. Tissue donor to host compatibility is important. In the instance of os and cartilage transplantation, fresh or frozen os graft is ordinarily dead on transplantation unless there is good grafted vascular supply. Over time, expressionless bone is replaced past living bone. In the case of cortical bone this process is slow and may simply involve a few millimeters of depth. Tissue donor to host compatibility is a minor issue unless marrow elements are present. Unfrozen articular cartilage has chondrocyte survival, no rejection, and no posterior graft cell repopulation (chimerism). Although the graft articular cartilage matrix seems to be maintained over years, at that place are studies of cartilage metabolism and allogenic osteochondral grafts that show a lower cell count and reduce metabolism. The os is somewhen replaced by host bone but the bone of the cartilage bone interface does not participate in this procedure [50]. Brighton et al., examined medicines of articular cartilage storage with the application of tissue culture techniques. Mechanical and histologic examination of the stored constructs was normal when compared with controls [51]. I performed some other report in which the osteo-articular constructs were stored for thirty days, was again found that the biomechanical properties of the constructs were essentially normal when compared with controls. Histologic examination evidence that there was merely pocket-sized swelling of the chondrocytes and that the histochemistry appeared to be normal. Incorporation of SO4 seem to exist increased when compared with fresh command samples simply there appeared to be normal amounts of hydroxproline and hexosamine. Animal studies were and then undertaken with transplantation of the stored osteoarticular allograft plugs into New Zealand White rabbits. The results were evaluated at 12 months after transplantation with gross examination every bit well as mechanical and biochemical testing. Over 70% of the transplanted osteoarticular grafts showed loss of cartilage thickness, irregularity, and fibrillation. Histologically, several of the graft showed show of an immune reaction with large numbers of inflammatory cells and apparent graft necrosis. At that place was only l% retention of proteoglycan content but hydroxyproline content was not significantly unlike from control values. Only twenty% of the grafts appeared to accept preserved matrix integrity. Gel filtration chromatography of sulfate- labeled macromolecules in guanidine HCL was used to index the quality of the proteoglycans synthesize by fresh and stored osteoarticular allograft plugs. A characterization of the proteoglycan eluted from the grafts revealed that the stored grafts had partially degraded molecules despite a normal staining with Safranin-O and normal SO4 content. This qualitative degeneration of cartilage cardio glycan was idea to resultant susceptibility to mechanical an immunologic failure. Amiel and Schacher have also looked at cartilage storage [52,53].
Osteochondral allografts can be of three types: Shell allograft, mosaicplasty, and large fragment osteochondral allograft. Fresh allograft replacement surgery has been performed for master osteoarthritis, osteonecrosis, osteochondritis dissecans, and posterior medical defects. Procedures for protuberant fresh allograft material have been established by the American Clan of tissue banks. Donors must exist beneath xxx years of historic period which is not to maximize cartilage quality. The grafts are harvested under strict aseptic conditions and usually inside 24 hours after death. The entire articulation is harvested with joint capsule ligaments cartilage and metaphyseal bone beingness excised en bloc. Cultures are taken after harvest in the entire joint is immersed in lactated Ringer's solution with cefazolin and bacitracin antibiotics beingness included in the solution. Later on the container sealed off, it is stored at iv°C. Histocompatibility markers are not obtained. Donor and recipient are matched for size and transplantation must take place in less than i week.
A biomechanical and biochemical study conducted past Jimenez and Brighton studied fresh frozen osteochondral allografts and rabbits [44]. The graft was stored for 30 days and tissue cultures weather condition. They establish that 70% to 75% of the sample did not announced normal and manifested early degenerative changes including fibrillation and loss of proteoglycan. By comparing cryopreservation techniques with fresh osteochondral allograft, it was constitute that cryopreservation resulted in less immunologic antigenicity but also marked degeneration of the cartilages matrix and chondrocyte viability. In 1985 study past Czitrom et al., found that chondrocyte viability and fresh osteochondral allograft ranged from 69% to 99% when assessed in culture media from biopsy specimens by S04 and 3H-cytidine auto radiography and grafts i-6 years after implantation [54]. One six-twelvemonth graft had only 37% chondrocyte viability but they were actively producing proteogylcans. Human chondrocytes in the cartilage of a fresh osteochondral graft tin can apparently survive prolonged periods of fourth dimension. Oakshott et al., analyzed eighteen cases of failed fresh allografts and found that the grafted bone had indeed died [55]. Twelve of the xviii grafts had viable cells at 13-92 months. Convrey et al., has too found allograft chondrocyte survival viii years after implantation [56]. There seems to be a cell mediated immune response that is responsible for the allografted os graft rejection. Langer et al. has demonstrated a positive lymphocytic migration test afterward graft transplantation. The allograft may actually soften with subsequent creeping substitution from the posterior tissue taking place over several years. Graft subsidence has really been observed at 2-three years condition postindex surgery and is likely because of a delay in early revascularization as a consequence of the immune response and softened subchondral os. Bones animal research has shown the importance of isolation of allogenic chondrocytes from post subchondral bone [57] (Effigy 7).
Figure 7. Damage cartilage equally it appears on radiograph
Mohamed et al., followed 92 posttraumatic articulatio genus injuries who received fresh allograft reconstruction of a pocket-sized osteochondral lesion of either the tibia or femur [58]. Failure was defined as less than 20 points of improvement on the knee cess scoring system from hospital for special surgery, any revision procedure, or subsequent patient assessment that the human knee was worse. There was clinical success and 75% at five years, 64% to 10 years and 63% at 14 years after implantation (Figures viii and nine).
Effigy 8. Damaged cartilage
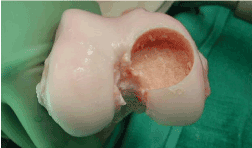
Effigy 9. Allograft recipient site
Man chondrocytes have been cultured for autologous chondrocyte transplantation and human since 1987. Peterson et al., reported on successful treatment of focal patellar defects in a rabbit mode with the use of transplanted cultured autologous chondrocytes [59]. The cultured chondrocytes were injected nether a periosteal flap which was sutured over the patellar defect. Examination 1 yr afterwards transportation revealed that a cartilage-like tissue covered about 70% of the defects. In 1994, Britberg et al. [60] reported on the results of 23 patients treated with Autogenous Chondrocyte Implantation (ACI) with a 16-66-month follow-up [sixty]. Second expect arthroscopy allowed clinical examination at 3 months. These examinations revealed that the transplants were level with the surrounding tissue and somewhat spongy when probed. A 2nd arthroscopic examination was performed on a subset of these patients at 12-46 months and clinical examination revealed that the transplant had progressed through a maturation process and had firmed upwardly. During the 2nd arthroscopic process, biopsy specimens extending to the subchondral bone were taken from the primal portion of the transplant. Histologic test and biochemical staining of the transplant biopsies revealed that the transplant had an abundance of type II collagen like to normal articular cartilage.
It has been theorized that considering chondrocytes are encased by matrix that they lack the power to migrate to the site of chondral injury and actively participate in a repair process [61,62]. And we'll studies have shown the partial-thickness chondral injuries can sometimes undergo repair by stem prison cell migration from adjacent synovial tissues. Full-thickness chondral lesions and developed humans have non shown the capacity to heal. Stem cell migration from the underlying subchondral bone with defect filling by fibrocartilage has been studied extensively and has produced variable results equally to effectiveness. The resulting fibrocartilage repair tissue is predominantly fibrous in nature, with variable numbers of chondrocytes present. The tissue at a microscopic level seems unorganized and lacks the biochemical and viscoelastic characteristics of normal Hylan cartilage [63]. The implanted chondrocytes, when studied with radioisotope labeling, seemed to physically adhere to the wound bed and significantly contribute to the repair tissue. Breinan et al., created four mm defects in the trochlear groove and 3 randomized groups of dogs [64]. 1 grouping had an empty defect, i grouping had periosteum sewn over the defect, and a throat group had autogenous chondrocyte transplantation with periosteum. Histomorphometric analysis at 12 and 18 months revealed the defect fill was 36% to 76%, Hylan-like cartilage was 10% to 23%, and integration with surrounding cartilage was xvi% to 32% with no significant divergence between three groups. Peterson et al., performed arthroscopic second looks on 65 patients who had undergone ACI during a 2 to 9 yr issue written report [65]. Quality of the repair tissue was assessed as to defect fill, integration with the surrounding cartilage, and surface mechanical characteristics. Biopsy specimens from the primal portion of the grafted site were also obtained. Macroscopic assessment of the repair tissue revealed that the implants slowly matured over the course of the yr. Arthroscopy performed in the showtime 2-3 months showed a repair construct that was typically soft and had a wavelike blueprint of graft motility when probed. Past 12 months the repair tissue was almost as firm every bit the next cartilage. Histopathologic analysis was performed on 37 biopsy specimens. 3 pathologists who had been blinded to treatment outcomes were asked to evaluate the tissue samples. Nearly of the specimens were thought to accept a homogenous matrix with depression cellularity, with rounded chondrocytes encased in typical lacunae, which was consequent with hyaline cartilage. The size, shape, and cell features were feature of hyaline cartilage. Immunohistochemical staining was positive for type II cartilage and normal proteoglycan content and all specimens that had a Hylan-similar appearance. At that place was a positive correlation between Hylan like Reconstruction of the defect and clinical outcome differences on histologic examination wore a greater number of chondrocytes with a more random distribution of cell colonies and absence of columnar organization. As a periosteal envelope was created to facilitate the constructs application, it should be noted that in some biopsy specimen the most superficial layer was composed of remnant fibrous sites which were likely incorporated from the periosteum. The periosteum seems to incorporate into the repair tissue through metaplasia and is mechanically debrided from the surface of the construct during the commencement 3-5 months. Contaminating cells may not come from just the periosteal envelope. Cells from the synovium and bone may also contribute biologically to the chondrocyte culture medium. Lovstedt et al., found that by using a combination of primers for ostial keelson and collagen types 2A+B, they were able to positively identify chondrocytes before cell culturing [66]. The culturing of chondrocytes for ACI has been time-dependent. Cell culture methods unremarkably required that the cell isolation have place within half dozen hours after surgery, followed past augmentation and implantation in 14-21 days. Leela and Bentley examined the capability of chondrocytes from damaged articular cartilage to role and ACI [67]. In an effort to avoid iatrogenic damage to the knee, the debrided cartilage in the perilesional area was collected and enzymatically digested and candy according to standard protocol. Histologic and immunohistochemical analysis was carried out as well as assays for DNA and GAGs. The chondrocytes obtained from the debrided cartilage lesion were equivalent to those obtained from harvested good for you cartilage areas. Sufficient cell numbers for implantation were achieved for all patients. The but departure was that the patients who had cartilage scavenged from large degenerative lesions required significantly longer times in civilisation to obtain the required number of cells.
Matrix associated autologous under site transportation/implantation is a new operation that uses chondrocytes seated into engineered matrix. it is using the treatment of localized full-thickness cartilage defects. Behrensa et al., published in knee on 5 year follow-up of 30 patient's treated with matrix associated autologous chondrocyte transplantation/implantation [68]. Four unlike scoring systems including the Meyer score, Tegner-Lyshlom activity score, Lysholm- Gillquist score, and the international cartilage repair Society score were used in the conjunction with a subset of patients who underwent biopsy. v years after implantation, 72% of patients rated the function of their knee much better than earlier (Figures 10 and eleven).
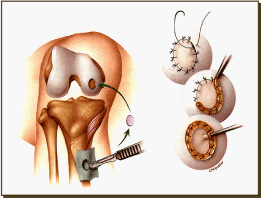
Figure 10. ACI Surgical procedure
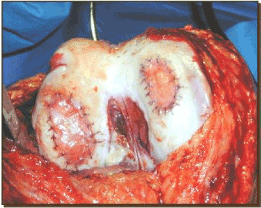
Figure eleven. Status mail service ACI implantation
The innovation of cell-based therapies to care for articular cartilage defects began with an autologous chondrocyte implantation (ACI) technique start described over 25 years ago [69,lxx]. Early on generations of ACI utilized an injectable suspension of cultured chondrocytes beneath a secured manufactured biologic embrace over the defect site [69,seventy]. First generation of ACI used a porcine periosteal comprehend (ACI-P) which was replaced past a collagen type I/III cover in the second generation ACI-C both of which were secured with sutures; the collagen cover was proven to have lower instances of post-operative hypertrophy compared to periosteal covers [69,seventy]. Third generation of prison cell-based regenerative therapy for treatment of total-thickness cartilage defects is referred to as maxtrix-induced autoloug chondrocyte implantation (MACI) which adapted the second generation collage type I/Three membrane into a bilayer membrane "seeded" with autologous chondrocytes and secured with fibrin glue [69-74]. MACI is a two-stage process including harvesting of chondrocytes for graft preparation so transplant/implantation of chondrocyte matrix graft approximately 4-vi weeks later. Chondrocytes are isolated and cultured in a mono layer. However, chondrocytes dedifferentiate in monocultures and restoration of their chondrocytic phenotype is disquisitional to cartilage graft success (Figures 12 and 13) [73].
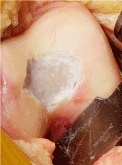
Figure 12. Damaged cartilage
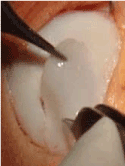
Figure 13. Allograft implant
Os Marrow Lesions (BML) are associated with mechanical or inflammatory injury to subchondral bone and are identifiable past a distinct MRI water signal. All-time visualized on T2 fat suppressed MR imaging, BML are recognized every bit hyperdense homeogenetic water signals of subchondral os without sharp margins or respect for anatomical boundaries. Although nigh commonly occurring in load-bearing regions, BML location has also been correlated with entheses in inflammatory cases. In prior literature, BMLs were referred as os marrow edema due to their feature MRI water signal. The term os marrow lesion has been adopted subsequently histological analysis revealed BML lack edematous changes; BML histology has been described as fibrosis lymphocytic infiltrates and increased vascularization of subchondral bone. The size of BML have proven to be clinically pregnant and positively correlates with illness progression and pain [75]. It is well-known that knee joint space narrowing appreciated on plainly x-rays inconsistently correlate to the severity of symptoms reported by patients. Notwithstanding, the presence of BML visualized on MRI strongly correlated with severity of reported hurting. Bone marrow edema is postulated to contribute to the progression of cartilage defect/arthropathy because the compromised bone fails to distribute force laterally through cartilage and the softer tissues receives focal, vertical forces. Chronic BML associated with OA stand for a new target for treatment though, adapted, but familiar techniques of internal fixation and os stimulation utilize to treat not-healing fractures. The role of subchondroplasty is to deliver a bone substitute to the subchondral BML to restore structural back up and facilitate bony healing [76]. BML associated with OA are hypothesis to develop through an "inside-out" mechanism; whereby microfractures elicit localized proinflammatory cytokines and vascoactive agents in bone marrow. In contrast to an "outside-in" machinery characteristic of inflammatory arthropathies such as RA or psoriasis whereby inflammatory cells enter into the bone marrow [75].
In 2007 the minimally invasive orthobiologic procedure, Subchondroplasty (SCP), was developed and has been utilized to treat chronic and astute Bone Marrow Lesions (BML) of the subchondral bone. Nether fluoroscopic guidance a flowable bone substitute is injected direct into the BML until a darkened blush is seen to mimic the BML design nether fluoroscopy and is ofttimes combined with additional arthroscopic and extra-articular procedures [77]. The properties of the injectable CaP are important to consider. This synthetic bone substitute must be injectable and flowable but solidify via endothermic reaction as to avert surrounding prison cell death. As a solid the CaP needs have similar structural properties to native cancellous bone to minimize inflammatory response. Lastly, the bone substitute must be biologically reabsorbable to permit for bony healing [78]. This scaffolding-like os substitute facilitates the remodeling capabilities of local osteoclasts or osteoblasts [79]. For patients with notable knee misalignment, greater than 8 degrees of varus or valgus malalignment, it is strongly recommended that SCP is paired with articulation realignment procedures. SCP have frequently been documented to exist paired with arthroscopic procedures every bit needed after intra-articular pathologies are assessed. Thus far there have been minimal complications associated with SCP. A few patients accept experienced persistent post-operative hurting due to bone substitute hardening in surrounding soft tissue. Patients who later on elected to proceed to definitive handling of TKA did not experience any complications due to synthetic os substitute [78]. The subchondroplasty procedures has primarily been described to care for BML of the tibiofemoral articulation. More than contempo studies accept introduced the SCP procedure for treatment of BML of the patellofemoral articulation. SCP have been well-nigh successful in treating BML of load-bearing regions of the knee in contrast to treating BML at entheses [79]. While subchondroplasty studies have revealed promising results for this procedure, the limits to which this process can provide patients with pain relief and improved function are still beingness adamant. Chatterjuu et al., provided evidence that injectable CaP into subchondral BML did not reliably provide hurting relief and/or improved function to patients with advanced knee OA [lxxx]. This written report demonstrates that 31.eight% of patients, out of 22 patient study grouping, with grade Iii or IV cartilage defects experienced a poor surgical outcome which was measured mail-operatively with KOOS, Tegner and Lysholm scores [80]. The 6-yr prospective nonrandomized study of subchondroplasty effectiveness published past Cohen and Skarkey reported durable improvement of pain scores and symptom/role scores from patients status post genu subchondroplasty via arthroscopy [77]. Of note, patients with grade IV tricompartmental knee OA were excluded from this written report group. Afterwards undergoing SCP, 70% of the included patients who were originally consulted for total knee arthroplasty did not go along to this definitive treatment option within the study period. Interestingly, those patients who proceeded onto TKA did so within the commencement yr after SCP. The advantages of subchondroplasty are notable for its minimally invasive approach that preserves native joint infinite and provides reliable pain relief in the short and long term with a shorter postoperative recovery compared to TKA [77].
Osteotomy of the tibia or femur may be performed to amend the mechanical axis of involved knee joints (Effigy fourteen-sixteen)
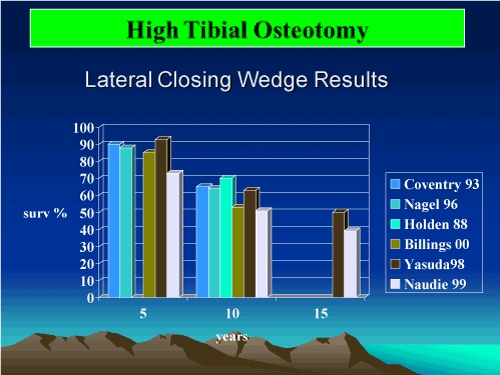
Figure fourteen. Loftier Tibal Osteotomy

Figure fifteen. Supracondylar varus osteotomy
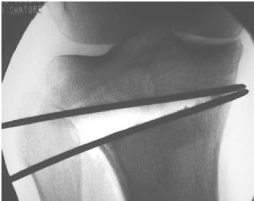
Figure 16. Loftier tibial osteotomy
Orthopedic surgeons involved and cartilage replacement and preservation surgery must exist aware of many outstanding basic biologic problems they face. Articular cartilage and its adjoining bony surface is a remarkable organ that demands respect for its biology and crave attention to the many mechanical and biochemical details that should exist considered for surgical planning. It is too incumbent on the surgeon to go along abreast of changes in the biologic literature and evidence pain clinical literature that will continually alter their practice. Although cartilage has a highly stable component to its matrix, it is capable of recovery and prolonged life given the proper surroundings.
- Goldring MB, Goldring SR (2007) Osteoarthritis. J Cell Physiol 213: 626-634. [Crossref]
- Moreland LW (2003) Intra-articular hyaluronan (hyaluronic acid) and hylans for the treatment of osteoarthritis: mechanism of action. Arthritis Res Ther 5: 54-67. [Crossref]
- Wesseling J, Welsing PM, Bierma-Zeinstra SM, Dekker J, Gorter KJ, et al. (2013) Impact to cocky-reported comorbidity on physical and mental health status in early symptomatic osteoarthritis: The Check (Cohort hip and Cohort knee) study. Rheumatology (Oxford) 52: 180-188. [Crossref]
- Hagans MP, Cederberg H, Mikkola I, et al. (2012) Reduction and metabolic syndrome, obese young Mammon and associated with exercise-induced body limerick changes during military machine service. Diabetes Res Clin Pract 98: 312-319.
- Costa FF, Montenegro VB, Lopes TJ, Costa EC (2011) Combination of take a chance factors for metabolic syndrome in the military machine personnel of the Brazilian Navy. Arq Bras Cardiol 97: 485-492. [Crossref]
- Bay-Jensen AC, Hoegh-Madsen Due south, Dam E, Henriksen K, Sondergaard BC, et al. (2010) Which elements are involved in reversible and irreversible cartilage degeneration in osteoarthritis? Rheumatol Int xxx: 435-442. [Crossref]
- Goldring MB, Otero M (2011) Inflammation in osteoarthritis. Curr Opin Rheumatol 23: 471-478. [Crossref]
- Maly MR (2008) Abnormal and cumulative loading in knee osteoarthritis. Curr Opin Rheumatol 20: 547-552. [Crossref]
- Bagga H, Burkhardt D, Sambrook P, March L (2006) Longterm effects of intraarticular hyaluronan on synovial fluid in osteoarthritis of the knee. J Rheumatol 33: 946-950. [Crossref]
- Caligaris M, Canal CE, Ahmad CS, Gardner TR, Ateshian GA (2009) Investigation of the frictional response of osteoarthritic human being tibiofemoral joints and the potential beneficial tribological effect of healthy synovial fluid. Osteoarthritis cartilage 17: 1327-1332. [Crossref]
- Murphy L, Schwartz TA, Helmick CG, Renner JB, Tudor G, et al. (2008) Lifetime risk of symptomatic knee joint osteoarthritis. Arthritis Rheum 59: 1207-1213. [Crossref]
- Rivera JC, Wenke JC, Buckwalter JA, Ficke JR, Johnson AE (2012) Posttraumatic osteoarthritis acquired by Battlefield injuries: The primary source of disability and Warriors. J Am Acad Orthop Surg 20: 64-69. [Crossref]
- Lohmander LS, Englund PM, Dahl LL, Roos EM (2007) The long-term outcome of inductive cruciate ligament and meniscus injuries: osteoarthritis. Am J Sports Med 35: 1756-1769. [Crossref]
- Hoshino Y, Fu FH, Irrgang JJ, Tashman S (2013) Tin joint contact dynamics be restored past anterior cruciate ligament reconstruction? Clin Orthop Relat Res 471: 2924-31.
- Jones JC, Burks R, Owens BD, Sturdivant RX, Svoboda SJ, et al. (2012) Incidence and risk factors associated with meniscal injuries among active-duty US military service members. J Athl Railroad train 47: 67-73. [Crossref]
- Ding L, Heying E, Nicholson Due north, Stroud NJ, Homandberg GA, et al (2010) Mechanical touch on induces cartilage degeneration via mitogen activated poly peptide kinases. Osteoarthritis cartilage 18: 1509-17. [Crossref]
- Jaquet GJ (2013) EWI VIII: Sequelae of combat injuries. AAOS 2022; April. Available at www.AAOS.org\news\AAOS now\apr13\clinical 2.asp; accessed viii July 2022.
- Goldberg VM, Goldberg Fifty (2010) Intra-articular hyaluronans: the treatment of knee pain in osteoarthritis. J Pain Res 3: 51-56. [Crossref]
- Genda Due east, Li Thou, Barrance PJ (1996) Functional analysis of hip joint contact pressure. Trans Orthop Res Soc 21: 416.
- Kitaoka HB, Kura H, Luo ZP (1996) Contact feat2021 Copyright OAT. All rights reservthop Res Soc 21: 396.
- Ameye L, Immature MF (2002) Mice deficient in small leucine-rich pro, normal in vivo models for osteoporosis, osteoarthritis, Ehkers-Dabkins syndrome, muscular dystrophy and corneal disease. Glycobiology 12: 107R-116R. [Crossref]
- Tsumura H, Miura H, Iwamoto Y (1998) Three-dimensional pressure distribution of the man hip joint comparison between normal hips and dysplastic hips. Fukuoka Igaku Zasshi 89: 109-118. [Crossref]
- Walker PS, Erkman MJ (1975) The role of the menisci in force transmission beyond the human knee. Clin Orthop Relat Res 184-192. [Crossref]
- Brown TD, Shaw DT (1984) In vitro contact stress distribution on the femoral condyles. J Orthop Res 2: 190-199. [Crossref]
- Buckwalter JA, Einhorn TA, Simon SR (2000) Orthopedic bones scientific discipline. American cadmium orthopedic surgeon, Chicago.
- Haskell VC, Sid Jara JW (1970) Physical backdrop N Polley disperse city of protocol I tin from bovine nostril cartilage. J Biol Chem 245: 4920-4930.
- Schinagl RM, Gurskis D, Chen Air conditioning, Sah RL (1997) Depth-dependent confined compression modulus of full-thickness bovine articular cartilage. J Orthop Res 15: 499-506. [Crossref]
- Wong M, Wuethrich P. Buschmann MD, Eggli P, Hunziker E (1977) Chondrocyte Biosynthesis correlates with local tissue strain in static compressed developed articular cartilage. J Orthop Res fifteen:189-196. [Crossref]
- Cochran GVB (1982) A Primer of Orthopedic Biomechanics. New York: Churchill Livingston, pp. 413. [Crossref]
- Dye SF (1996) The knee as a biologic transmission with an envelope of function: a theory. Clin Orthop Relat Res 10-xviii. [Crossref]
- Goldenberg VM, Kuettner KE. Osteoarthritic disorders. Chicago: American Academy orthopedic surgeons; 1995: xxii-xxiii
- Martin JA, Scherb MB, Lembke LA, Buckwalter JA (2000) Impairment control mechanisms in articular cartilage: the role of the insulin-like growth factor I centrality. Iowa Orthop J 20: ane-10. [Crossref]
- Martin JA, Scherb MB, Lembke LA, Buckwalter JA (2000) Damage command mechanisms in articular cartilage: the role of the insulin-like growth gene I axis. Iowa Orthop J 20: 1-ten. [Crossref]
- Convery FR, Akeson WH, Keown GH (1972) The repair of big osteochondral defects. An experimental study in horses. Clin Orthop Relat Res 82: 253-262. [Crossref]
- Fuller JA, Ghadially FN (1972) Ultrastructural observations on surgically produced partial thickness defects in articular cartilage. Clinc Orthop 86: 193-205. [Crossref]
- Caplan AI (1991) Mesenchymal stem cells. J Orthop Res 9: 641-650. [Crossref]
- Rodrigo JJ, Steadman JR, Sillman JF (1994) Improvement of total thickiness chondral defect healing in the human subsequently debridement and microfracture using continuous passive motion. Am J Knee Surg vii: 109-116.
- Steadman JR, Rodkey WG, Singleton SB (1997) Microfracture technique and clinical results. Oper Tech Orthop vii: 300-304.
- Salter RB, Simmonds DF, Malcolm BW, Rumble EJ, MacMichael D, et al. (1980) The biological effect of continuous passive motion on the healing of total-thickness defects in articular cartilage. An experimental investigation in the rabbit. J Bone Joint Surg Am 62: 1232-1251. [Crossref]
- Shapiro F, Kiode Due south, Glimcher MJ (1994) Cell origin and differentiation in repair of fullthickness degevys of articular cartilage. J Bone Joint Surg 74: 523-533. [Crossref]
- Wakitani S, Goto T, Pineda SJ, et al. (1994) Mesenchymal cell based repair of larger, total thickeness defects of articular cartilage. J bone Joint Surg 76: 579-592. [Crossref]
- Lexer EL (1925) Articulation transplantation and arthroplasty. Urg Gynecol Obstet 40: 782.
- Lexer EL (1908) Substiution of whole or one-half joints from freshly amputated extremities past free plastic operation. Surg Gynecol Obstet half-dozen: 601.
- Jimenez SA, Brighton CT (1983) Experimental studies on the fate of transplanted articular cartilage. In: Friedlander GE, Mankin HJ, Sell KW (eds.) Osteochondral Allografts, cyberbanking and clinical applications. Boston: Brown, 73-79.
- Lane JM, Brighton CT (1977) Joint resurfacing in the rabbit using an autologous osteochondral graft: a biochemical and metabolic study of cartilage viability. J Bone Joint Surg 59: 218.
- Lipton MA (1978) Allotransplantation: a historical, biochemical, and biomechanical study. Trans Orthop Res Soc three: 12.
- De Nubile NA (1980) Allotransplantation of preserved and fresh articular cartilage in an osteochondral plug model. Trans Orthop Red Soc v: 74.
- Hangody L, Kisk Grand, Karpati Z (1992) Autogenous osteochondral graft technique for replacing human knee cartilage defects in dogs. Int Orthop v: 175-181.
- Hangody L, Kisk G, Karpati Z (1997) Autogenous osteochondral graft technique for replacing human knee cartilage defects in dogs. Int Orthop 5: 175-181.
- Nelson FR (1992) Chondrocyte technology: in search of articular cartilage. MUSC Orthop J two: 26-31. [Crossref]
- Brighton CT, Shadle CA, Jimenez SA, Irwin JT, Lane JM, et al. (1979) Articular cartilage preservation and storage I. Awarding of tissue civilization techniques to the storage of viable articular cartilage. Arthritis Rheum 22: 1093-1101. [Crossref]
- Oates KM, Chen Air-conditioning, Young EP, Kwan MK, Amiel D, et al. (1995) Effect of tissue culture storage on the in vivo survival of canine osteochondral allografts. J Orthop Res 13: 562-569. [Crossref]
- Stevenson Due south, Dannucci GA, Shrkey NA, Pool RR (1989) The fate of articular cartilage after transplantation of freash and cryopreserved tissue-antigen-matched and mismatched osteochondral allografts in dogs. J Os Articulation Surg 71: 1297-1307. [Crossref]
- Czitrom AA, Keating S, Gross A (1990) The viability of articular cartilage in fresh osteochondral allografts after clinical transplantation. J Os Joint Surg 72: 574-581. [Crossref]
- Oakeshott RD, Farine I, Pritzker KP, Langer F, Gross AE (1988) A clinical and histologic assay of failed fresh osteochondral allografts. Clin Orthop Relat Res 283-294. [Crossref]
- Convery FR, Akeson WH, Amiel D, Meyers MH, Monosov A (1996) Long-term survival of chondrocytes in an osteochondral articular cartilage allograft. A example report. J Os Joint Surg Am 78: 1082-1088. [Crossref]
- Moskalewski S, Hyc A, Osiecka-Iwan A (2002) Immune response past host later on allogeneic chondrocyte transplant to the cartilage. Microsc Res Tech 58: 3-13. [Crossref]
- Mohamed MN, Beaver RJ, Gross AE (1992) The long-term success of fresh, small fragment osteochondral allografts used for intra-articular post-traumatic defects in the knee joint. Orthopaedics 15: 1191-1199. [Crossref]
- Peterson L, Menche D, Grande D (1984) Chondrocytes transplantation: an experimental model in the rabbit. Trans Ortho Res Soc 18: 218.
- Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, et al. (1994) Handling of deep cartilage defects in the knee joint with autologous chondrocyte transplantation. N Engl J Med 331: 889-895.
- Hirotani H, Ito T (1975) Chondrocyte mitosis in the articular cartilage of femoral heads with various diseases. Acta Orthop Scand 46: 979-986. [Crossref]
- Rothwell AG, Bentley G (1973) Chondrocyte multiplication in osteoarthritic articular cartilage. J Bone Joint Surg Br 55: 588-594. [Crossref]
- Grande DA, Pitman MI, Peterson 50, Menche D, Klein M (1989) The repair of experimentally produced defects in rabbit articular cartilage by autologous chondrocyte transplantation. J Orthop Res 7: 208-218. [Crossref]
- Breinan HA, Minas T, Hsu HP, Nehrer S, Sledge CB, et al. (1997) Effect of cultured autologous chondrocytes on repair of chondral defects on a canine model. J Os Joint Surg 79: 1439-1451. [Crossref]
- Peterson 50, Minas T, Brittberg M, Nilsson A, Sjögren-Jansson Eastward, et al. (2000) Two- to 9-year outcome after autologous chondrocyte transplantation of the articulatio genus. Clin Orthop Relat Res 212-234. [Crossref]
- Lovstedt One thousand, Thornemo One thousand, Lindahl A (2002) Identification of Contaminating Cells from Biopsies Used for Articular Cartilage Repair. Toronto: International Cartilage Repair Club, 73.
- Leela CB, Bentley 1000 (2002) A novel Source of Cells for Autologous Chondrocyte Transplantation in the Adult Articulatio genus. Toronto: International Cartilage Repair Society, 54.
- Behrens P, Bitter T, Kurz B, Russlies M (2006) Matrix-associated autologous chondrocyte transplantation/implantation (MACT/MACI) - v yr follow upwardly. Knee 13: 194-202. [Crossref]
- Bartlett Westward, Skinner JA, Gooding CR, Carrington RWJ, Flanagan AM, et al. (2005) Autologous chondrocyte implantation versus matrix-induced autologous chondrocyte implantation for osteochondral defect of theknee. J Os Articulation Surg Br 87-B: 640-645. [Crossref]
- Basad E, Wissing FR, Fehrenbach P, Rickert Chiliad, Steinmeye J, et al. (2015) Matrix-induced autologous chondrocyte implantation (MACI) in the knee: clinical outcome and challenges. Knee joint Surg Traumatol Arthrosc 23: 3729-3735. [Crossref]
- Cherubino P, Grassi FA, Bulghrtoni P, Ronga M (2003) Autologous chondrocyte implantation bilayer collagen membrane: A preliminary written report. J Orthop Surg (Hong Kong) 11: x-15. [Crossref]
- Ebert JR, Fallon One thousand, Smith A, Janes GC, Wood DJ (2015) Prospective clinical and radiographic evaluation of patellofemoral matrix-induced autologous chondrocyte implantatio. Am J Sports Med 43: 1362-1372. [Crossref]
- Brittberg M (2010) Cell Carriers as the Next Generation of Prison cell Therapy for Cartilage Repair: A Review of the Martrix-induced Autologous Chondrocyte Implantation Procedure. Am J Sports Med 38: 1259-1271. [Crossref]
- Marlovits South, Aldrian Due south, Wondrasch B, Zak Fifty, Albercht C, et al. (2012) Clinical and Radiographic Outcomes 5 Years Afterwards Matrix-Induced Implantation in Patients With Symptomatic, Traumatic Chondral Defects. Am J Sports Med 20: one-viii. [Crossref]
- Eriksen EF, Ringe JD (2012) Bone marrow lesions: a universal bone response to injury? Rheumatol Int 32: 575-584. [Crossref]
- Sharkey PF, Cohen SB, Leinberry CF, Parvizi J (2012) Subchondral bone marrow lesions associated with knee osteoarthritis. Am J Orthop (Belle Mead NJ) 41: 413-417. [Crossref]
- Cohen SB, Sharkey PF (2016) Subchondroplasty for Treating Bone Marrow Lesions. J Articulatio genus Surg 29: 555-563. [Crossref]
- Cohen SB, Sharkey PF (2012) Surgical treatment of osteoarthritis pain related to subchondral bone defects or bone marrow lesion: subchondralplasty. Tech Knee Surg 11: 170-175.
- Farr J 2, Cohen SB (2013) Expanding applications of the subchondroplasty procedure for the treatment of bone marrow lesions observed on magnetic resonance imaging. Oper Tech Sports Med 21: 138-143.
- Chatterjuu D, McGee A, Strauss E, Youm T, Jazrami L (2015) Subchondral calcium phosphate is ineffective for bone marrow edema lesions in adults with advanced osteoarthritis. Clin Orthop Relat Res 473: 2334-2342. [Crossref]
Editorial Data
Editor-in-Chief
Martin Grabois
Baylor College of Medicine
Commodity Type
Review Commodity
Publication history
Received engagement: May 28, 2022
Accustomed date: July 10, 2022
Published appointment: July 13, 2022
Copyright
© 2022 Pettit T. This is an open up-admission article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted apply, distribution, and reproduction in any medium, provided the original author and source are credited.
Commendation
Pettit T (2017) Cartilage damage: a review of surgical repair options and outcomes. Phys Med Rehabil Res 2: DOI: 10.15761/PMRR.1000147

Figure i. Damaged cartilage of femur

Effigy 2. Handling algorithm for cartilage repair

Figure three. Microfracture of exposed os

Effigy four. Histologic view of microfracture

Figure 5. Mosaicplasty

Effigy 6. Repair tissue from microfracture

Figure seven. Damage cartilage every bit it appears on radiograph

Figure 8. Damaged cartilage

Effigy 9. Allograft recipient site

Figure 10. ACI Surgical process

Figure 11. Status post ACI implantation

Figure 12. Damaged cartilage

Effigy thirteen. Allograft implant

Figure 14. Loftier Tibal Osteotomy

Figure 15. Supracondylar varus osteotomy

Effigy 16. High tibial osteotomy
Source: https://oatext.com/cartilage-damage-a-review-of-surgical-repair-options-and-outcomes.php
Posted by: sengerarte1978.blogspot.com


0 Response to "What Is The Term For Surgical Repair Of Cartilage?"
Post a Comment